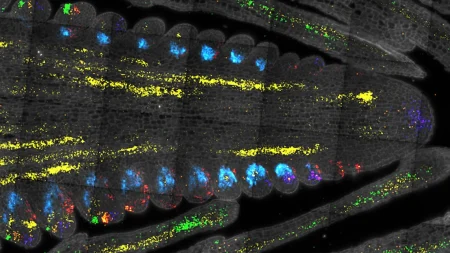
Biomedical engineers at Duke University have developed a novel method to better understand and test treatments for rare muscle disorders known as dysferlinopathy or limb girdle muscular dystrophies 2B (LGMD2B). This technique involves growing complex, functional 3D muscle tissue from stem cells in the laboratory, which creates a platform that mimics patient symptoms and responses to treatment. In their initial study, researchers exposed some of the biological mechanisms behind the loss of mobility characteristic of LGMD2B and demonstrated that a combination of existing therapies may help alleviate some of the severe symptoms associated with the disease.
LGMD2B is a rare condition that affects about eight individuals per million globally. Unlike the more common Duchenne muscular dystrophy, LGMD2B affects both men and women, generally appearing later in life, and while it is rarely fatal, it leads to significant weakness in the legs and shoulders, often necessitating the use of wheelchairs. The disease is caused by a genetic disorder that prevents the body from producing a fully functional form of the dysferlin protein, with no currently approved treatments available. The multifunctional dysferlin protein is responsible for activities such as sealing muscle membrane holes, regulating calcium balances essential for muscle contractions, and controlling cellular metabolism.
Researchers have run into challenges in understanding and treating LGMD2B due to the mild symptoms displayed by mouse models, which do not accurately reflect the disease’s impact on patients. Mice with the same genetic mutation as LGMD2B patients do not exhibit severe symptoms until almost a year into their two-year lifespan, hindering research progress. To address these challenges, Duke University scientists have utilized an engineered muscle platform that allows them to study the effects of dysferlin on skeletal muscle specifically, without interference from other cell types or altered blood metabolite profiles.
By maturing induced pluripotent stem cells derived from LGMD2B patients into muscle fibers, researchers have been able to observe and analyze a range of issues similar to those seen in patients. The lab-grown muscle cells displayed calcium handling deficiencies, leading to weaker contractions due to a loss of calcium reserves. Additionally, muscle damage went unrepaired in the absence of dysferlin, and an inability to burn fatty acids for energy production contributed to fat accumulation within the muscle fibers. Drug candidates dantrolene and vamorolone were tested in the study and showed promise in preventing calcium leaks, enhancing muscle strength, and reducing fat accumulation, highlighting their potential to treat LGMD2B.
Moving forward, the research team plans to incorporate immune and fat cells into their experiments to enhance complexity and better understand disruptions to cellular metabolism. They aim to identify new drugs that can address all aspects of muscle weakness, repair deficits, and metabolic issues in patients with LGMD2B. By expanding their system to include multiple cell types, researchers hope to unravel the root causes of muscle degeneration in these patients fully. The study was supported by the National Institutes of Health and the Jain Foundation, which focuses on finding a cure for LGMD2B.